Editor’s note: This text-based course is a transcript of the webinar, The Role of Resilience in Alzheimer's Dementia, presented by Teresa Fair-Field OTD, OTR/L.
Learning Outcomes
- After this course, participants will be able to list the top 5 resilience factors with an impact on cognitive health.
- After this course, participants will be able to identify the evidence-based best approach to exercise for older adults with dementia.
- After this course, participants will be able to recognize which sleep factors link to dementia in recent literature.
Introduction
Today, we will be talking about all the factors that we should be doing for ourselves as prevention and for our clients that are in the early phases of dementia to potentially change their disease course. You may have seen several articles come across and perhaps wondered about some of the studies behind these findings. We will talk about some of those today.
Current Scope of Dementia
- Alzheimer’s dementia
- Vascular dementia
- Lewy body dementia (DLB)
- Frontotemporal dementia (FTD)
- Normal-pressure hydrocephalus
- Mixed dementia
- Syndrome related dementias
We will be focusing exclusively on Alzheimer's dementia today, but there are several dementia types. Sometimes, you can see Alzheimer's dementia referred to as ADRD, Alzheimer's Dementia, and Related Dementias.
Vascular dementia occurs as a result of strokes or perhaps small TIAs. These may impact a person's judgment, problem-solving, and memory depending on what areas of the brain are affected. Based on its name, it is due to changes in vascularization of the brain tissue.
Lewy body dementia (DLB) results from micro formations of protein that occur in the cerebral cortex.
Frontotemporal dementia (FTD) is cellular damage in the frontal and temporal lobes of the brain. The frontal and temporal lobes are responsible for executive functioning. Executive functioning deficits more than memory affects things like inhibitory control, word-finding behavior, and some aspects of motor movement.
Normal-pressure hydrocephalus results from fluid built up in the brain's ventricles but without a measurable increase in intracranial pressure.
Mixed dementia is a combination of two types. The most common form of mixed dementia is that of Alzheimer's dementia that then gets overlaid with perhaps vascular dementia.
Of course, many neurological syndromes have accompanying dementia, such as Parkinson's disease. Parkinson's disease also contains Lewy bodies, though not everyone with Lewy body dementia has Parkinson's. Huntington's disease and Creutzfeldt-Jakob disease also have their dementia types.
Alzheimer's Disease Risk Factors
Again, we will be focusing on Alzheimer's during this presentation. It is the most common form of dementia, making up 60 to 80% of all dementias. In the absence of other co-occurring findings or diagnoses contributing to the cause of dementia, Alzheimer's is the most likely.
Age
The age of the individual is still the current best-known risk factor for Alzheimer's disease. Of course, not everyone who ages is diagnosed with Alzheimer's disease, and further not everyone who is diagnosed with Alzheimer's disease is equally affected. Most symptoms begin to appear after age 60, and your risk increases as you age. Early-onset Alzheimer's is diagnosed at ages below 60 and remains rare. Age-related Alzheimer's incidence rates are expected to double from their current numbers to about 13.9 million people by 2060. That is less than 40 years away. This number is between three and 4% of the expected population.
Racial Differences
There are racial disparities in Alzheimer's, as seen in Figure 1.
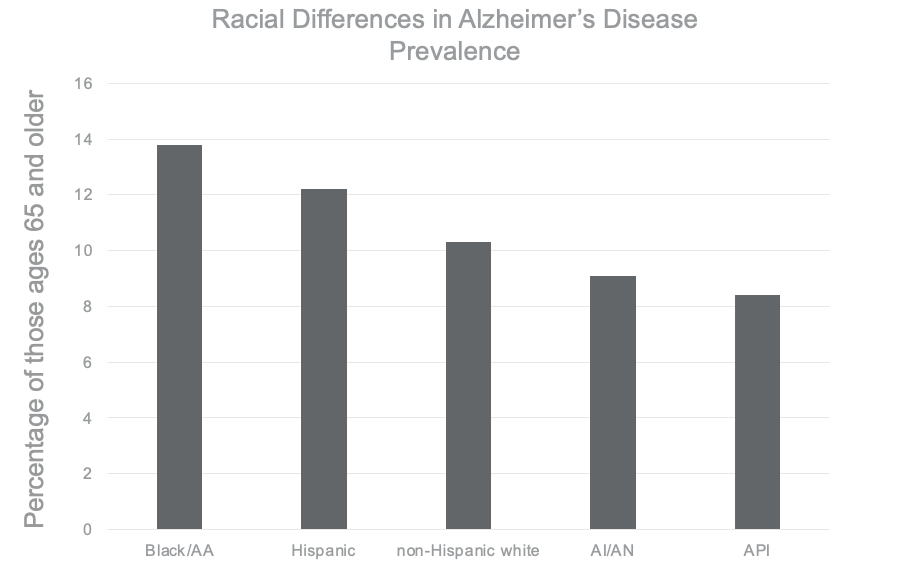
Figure 1. Bar graph showing racial disparities with Alzheimer's dementia. Click to enlarge the image.
African Americans have the highest incidence of Alzheimer's disease, with 13.8% of those ages 65 and older, followed by 12.2% in those identifying as Hispanic. 10.3% of those identifying as non-Hispanic White. 9.1% in the American Indian Alaska Native population (AIAN), and 8.4% in Asian and Pacific Islanders. A geographically and racially diverse team of more than 20 researchers examined factors affecting these Alzheimer numbers, and specifically the issue of reserve resilience and protective factors, and how those are exhibited in these racial and ethnic groups, as well as socioeconomic status (Barbour et al.). Be sure to look for that article in your references.
Identifying Alzheimer's Dementia
First, we need a basic understanding of some of the things researchers are using to identify Alzheimer's dementia.
- Vocabulary & Progression
- Plaques: abnormal clusters of protein fragments (β amyloid) that build up between nerve cells
- Tangles: twisted strands of (tau) protein that interrupts normal cellular communication
Some plaques are abnormal clusters of protein fragments. These are the beta-amyloid proteins that build up between the nerve cells. There are also tangles which are twisted strands of protein called tau, which interrupt normal cellular communication. It is important to note that both beta-amyloid and tau have purposeful functions in the healthy brain. Beta-amyloid becomes problematic only when abnormal levels of the protein begin to cause the formation of plaques. These plaques start to build up without getting cleared away out of the system.
Similarly, tau binds to and stabilizes the microtubules, which guides the flow of nutrients and molecules from the cell body down to the axons and dendrites. In Alzheimer's disease, abnormal changes occur, which cause tau to detach from those microtubules and then attach, creating a tangle. This tangle interrupts the flow the tau was supposed to manage.
Researchers are now learning that these mechanisms do not occur in isolation. Instead, it is an interplay of factors that causes the buildup of beta-amyloid. This buildup reaches a tipping point or what they call a cascade that causes tau to spread rapidly throughout the brain. The first phase or event of the disease appears to occur with a beta-amyloid cascade. Neuronal injury due to the tau protein seems to follow after that initial accumulation of beta-amyloid.
Any discussion of resilience and prevention needs to address the possibility of preventing this initial buildup of beta-amyloid.
Cognitive Reserve Theory
↑ Education, ↓ β-amyloid
- N=70 newly dx, Alzheimer’s disease
- Findings showed lower levels of β-amyloid in those with higher levels of education
- No relationship between education and tau
- Supports cognitive reserve theory “in the first period of clinical evolution”
- Findings progressively disappeared in advanced phases
Dumurgier, J., Paquet, C., Benisty, S., Kiffel, C., Lidy, C., Mouton-Liger, F., Chabriat, H., Laplanche, J-H., & Hugon, J. (2010). Inverse association between CSF Aβ 42 levels and years of education in mild form of Alzheimer's disease: The cognitive reserve theory. Neurobiology of Disease, 40(2), 456-459. https://doi.org/10.1016/j.nbd.2010.07.007
Cognitive reserve theories hypothesize that individual differences in brain health and in cognition can affect the expression and severity of Alzheimer's dementia. These findings are emerging in the evidence-based literature and represent the best current wisdom on the topic of resilience. Let's talk about these recent findings and what they might mean for your life and your practice.
The cognitive reserve theory suggests that at any level of severity, an individual with a higher cognitive capital could support more significant levels of brain pathology without demonstrating the same debilitating effects as someone with lower cognitive capital. This research dates back to the 1980s, when researchers completed autopsies of individuals with no apparent symptoms of dementia that were nonetheless found to have brain pathology consistent with advanced Alzheimer's disease.
Why is that? They theorized that they did not show symptoms because they had a large enough cognitive reserve to offset the debilitating effects of the damage. What besides post-mortem analysis could support that theory, and how has it progressed since that time? Several studies have emerged that examine the impact of education on beta-amyloid and tau proteins showing an inverse association. The inverse association is where one thing goes up, and another thing goes down.
This study looked at a sample of 70 subjects that were newly diagnosed with Alzheimer's disease. They performed a lumbar puncture to examine the cerebral spinal fluid of these individuals to look for the presence of beta-amyloid and tau. The result of their study showed an inverse association between education and β-amyloid, meaning when years of education went up, the presence of beta-amyloid went down. There was no such relationship between the level of education and the amount of tau.
However, as we talked about in the early phases of the disease, it is the beta-amyloid that we are watching. Their findings support the cognitive reserve theory in the first period of clinical evolution, meaning when Alzheimer's disease is first identified. Though the relationships seem to disappear in advanced stages of the disease, a few reasons are theorized why that may occur.
Brain Reserve Theory
Like this, brain reserve theories hypothesize that individual differences in the brain volume itself can affect the expression and severity of Alzheimer's disease.
Education & Brain Thickness
- N= 355
- 121 with AD, 121 with MCI, and 113 age-matched healthy controls
- Healthy controls = no significance
- Mild Cognitive Impairment = no significance
- Alzheimer’s disease group= greater cortical loss (supports cognitive reserve theory)
Liu, Y., Julkunen, V., Paajanen, T., Westman, E., Wahlund, L.-O., Aitken, A., Sobow, T., Mecocci, P., Tsolaki, M., Vellas, B., Muehlboeck, S., Spenger, C., Lovestone, S., Simmons, A., & Soininen, H. (2012). Education increases reserve against Alzheimer’s disease—evidence from structural MRI analysis. Neuroradiology, 54(9), 929–938. https://doi.org/10.1007/s00234-012-1005-0
It is hypothesized that a more robust volume of brain tissue one can support a more significant loss before reaching the threshold for clinical symptoms. A study on brain thickness was measured via structural MRI analysis in which brain segments were examined and compared with demographic and performance-based features of the subjects. These researchers based their hypothesis on years of schooling, but it was very disparate in the sample with six years of schooling plus or minus two years versus 13 years plus or minus two years. This study also controlled for things like intracranial volume, age, gender, country of origin, cognitive status, and multiple testing effects. There was no significant correlation between years of schooling and regional cortical thickness or volume in the age-matched healthy control group. However, some isolated sections of the brain were significantly larger in those with more education versus less. Overall, there were no significant findings amongst the control group. They also looked at individuals with mild cognitive impairment (MCI), often a diagnosis that precedes Alzheimer's disease. These subjects did not qualify for a diagnosis of Alzheimer's at this point, and there were no statistical differences in that group.
In the Alzheimer's group, they found that those with higher education rates did have more significant cortical loss than those with less education at the same functional level. Meaning, those individuals with more education may mitigate the effects of more significant cortical loss while retaining function. These findings also support reserve theory. However, these findings are not current, and conflicting research suggests that though these higher levels of education might mitigate the early stages of Alzheimer's disease, they could advance even more swiftly in the later stages, equalizing any early benefits.
Resilience
Improved Network Efficiency
- No relationship between education and CSF biomarkers (β-amyloid or tau proteins)
- Abnormal levels of β-amyloid or tau proteins were related to more efficient networks in the mild AD group!
- While education may not be protective, it may modify the effects of tau proteins in individuals with more highly efficient neural networks.
Weiler, M., Casseb, R. F., de Campos, B. M., de Ligo Teixeira, C. V., Carletti-Cassani, A. F. M. K., Vicentini, J. E., Magalhães, T. N. C., de Almeira, D. Q., Talib, L. L., Forlenza, O. V., Balthazar, M. L. F., & Castellano, G. (2018). Cognitive reserve relates to functional network efficiency in Alzheimer’s disease. Frontiers in Aging Neuroscience,10, 255–255. https://doi.org/10.3389/fnagi.2018.00255
Alzheimer's disease has not had a dramatic breakthrough, but they have benefited from various refinements in the research. One of those refinements is the progression of this cognitive reserve theory that now more closely resembles a model that we would call resilience. If we think about the brain being a resilient organ, this theory (most recent in 2018) supposes that cognitive reserve is less related to brain volume or the presence and absence of the identified proteins than how efficiently the brain works.
This group used a sample of 14 individuals with mild Alzheimer's disease, so at the front end of Alzheimer's, 28 with MCI, so those not yet diagnosed with Alzheimer's, and 28 controls. They found no relationship between education level and the amount of beta-amyloid or tau protein. They did find that education level was related to higher neural network efficiency in all groups. That is, people with more education and learning experiences essentially trained their brain's network. They found this true in the control group, the MCI sample, and the Alzheimer's disease sample. The act of training your brain increases its efficiency and more efficient neural networks based on their educational level.
Researchers also determined that abnormal beta-amyloid and tau proteins were related to more highly efficient networks in the sample with mild Alzheimer's. This is an exciting finding confirming that while education may not protect one from the onset of Alzheimer's, it may insulate the individual from the effects of pathology, creating resilience within the system. The brain then relies on its network efficiency during Alzheimer's effects occurring in the brain. It also opens the door to the potential benefit of continuing to provide cognitive stimulation to age-related cognitive decline on the front end of those already diagnosed with Alzheimer's. If we can continue to prompt those brains to access their neural efficient networks, this is exciting work for us as occupational therapists working in older adult and community health settings.
Another is the effect of reducing stress on resilience. As we know, our system becomes more resilient as we respond to stress, but reducing stress as much as we are able can improve the efficiency of those neural networks.
Reducing Stress
- Prolonged stress produced an ↑ level of cortisol associated with accumulation of tau
- Higher cortisol levels correlated with faster progression of the disease
Silva, M. V. F., Loures, C. de M. G., Alves, L. C. V., de Souza, L. C., Borges, K. B. G., & Carvalho, M. das G. (2019). Alzheimer’s disease: Risk factors and potentially protective measures. Journal of Biomedical Science, 26(1), 33–33. https://doi.org/10.1186/s12929-019-0524-y
Studies have found that prolonged stress produces an increased level of cortisol. Higher cortisol levels are associated with an accumulation of tau protein, so this correlates with a faster progression of the disease.
Protective Factors
Let's talk about the most exciting part, which is those factors that we would consider protective.
Cognitive Reserve
- Build cognitive flexibility
- Adopt a lifelong learning approach
- Take on new skills or tasks
- Build intergenerational relationships
Whatever the mechanism, cognitive reserve remains a solid protective factor. This is identified as the years of education because of the wide variability, but rather an adoption of a lifelong learning approach. You are training your brain to build that cognitive flexibility, and the more training opportunities you provide, the more neural efficiency. This embodies a lifelong learning approach.
How are you going to continue to access those neural pathways? One mechanism is to take on new skills or tasks, which we tend to be less inclined to do as we age. However, it may be the secret to tapping into this cognitive reserve function. Another is to build those intergenerational relationships that require our cognitive flexibility. When we work with younger individuals, this requires cognitive flexibility.
Physical Activity/Exercise
- Multiple sources cite physical fitness as a protective factor in cognitive health
- Active older adults have lower incidences of dementia
- While studies commonly cite aerobic fitness for its cardiovascular benefit, new findings support any sustained fitness and paired fitness having the greatest benefit
We have heard quite a bit about physical activity and exercise benefits via many studies and programs. Some we will be talking about in a moment regarding Alzheimer's, but multiple sources cite the benefits of physical fitness as a protective factor in cognitive health. There are many mechanisms by which this can occur. Some theorize that it is based on blood flow. We will talk more about it once we get into the area of sleep. We know that physical activity and exercise promote sleep. And, active older adults do have a lower incidence of dementia. While studies commonly cite aerobic fitness for its cardiovascular benefits, new findings support sustained and paired fitness as having the greatest benefit. Paired fitness includes aspects of both aerobic and strength-based exercise. Essentially, the best approach is the one the elder will perform consistently over time. Some studies have even explored dancing as physical fitness for the elderly. Thus, it is less important what the activity is and more about the ability to sustain it.
Environment
- Sensory programming
- Variability
- Indoors/Outdoors
The environment of the individual is also crucial for activating neural pathways. Many courses focus on sensory programming for individuals with Alzheimer's. It is particularly valuable to provide a varied environment and indoor and outdoor experiences for individuals with early Alzheimer's. Again, the environment may also affect sleep. If we think about indoors versus outdoors, we know that melatonin production decreases as we age. Any activities that stimulate melatonin production and exposure to sunlight have significant benefits as well.
Socialization
- Daily casual relationships may benefit the individual as much as friendships
- Unclear mechanism
- Effect on mood and connectedness
There are a handful of studies across that address socialization with this population. Daily casual relationships and socialization (at a fitness class. bookstore, or coffee shop) may have the same benefit as longstanding friendships as you age. It is not necessarily the quality of socialization that is necessary but rather the exposure to socialization that shows the benefit. This has an unclear mechanism, but the effect on mood and the individual's connectedness is significant.
Some studies show that elders with depression are more inclined towards Alzheimer's dementia as well. Although again, it is difficult to tease out which causes what, we know that those individuals engaged in social networks of some kind seem to be able to respond more resiliently to changes in cognition as they age.
Sleep
- Disrupted sleep and wakefulness lead to ↑ Aβ production and ↓ Aβ clearance
- Aβ accumulation results in disturbed sleep
- Acute sleep loss ↑ tau levels in CSF
- Chronic sleep loss accelerates tau spread in neural networks
- A significant relationship between sleep disturbances and Aβ aggregation
Sleep is one of my favorite topics and something I have addressed in another course on occupationaltherapy.com. Today, I will discuss the importance of sleep hygiene as we age because sleep research shows it has a powerful and unprecedented role in the cleanup of the brain. Without adequate sleep, the proteins discussed (Aβ, amyloid-beta, or also known as beta-amyloid) are cleared and ejected from the system. Disrupted sleep and wakefulness lead to an increase in beta-amyloid production and a decrease in beta-amyloid clearance. Thus, if you have disrupted sleep, it both causes a greater production of the protein as well as an accumulation.
Acute sleep loss also shows increased levels of tau in tested cerebral spinal fluid. Sleep has an impact on both proteins that are implicated in Alzheimer's disease. And, chronic sleep loss accelerates the spread of tau throughout the neural network. We now know that sleep can be one of the essential resilience functions in protecting against Alzheimer's disease or progression.
- Sleep Need
- “Changes in sleep efficiency and quality seem to precede the onset of cognitive decline in AD pts and progress in parallel with both cognitive dysfunction and the progression of AD pathology” (Wang & Holtzman, 2020, p. 105).
- >7 hours of sleep = lowest amyloid burden
- >6 to < 7 hours = intermediate amyloid burden
- < 6 hours = greatest amyloid burden
- “Participants who have preclinical amyloid pathology exhibit increased napping” (Wang & Holtzman, 2020, p. 106)
Sleep hygiene is something that we address as occupational therapists, and changes begin at home. Wang and Holtzman have written an excellent article that is cited in your references. If you are interested in all of their findings related to sleep, please seek out this article. It is very comprehensive and summarizes the results of sleep over time as well. Changes in sleep efficiency and quality seem to precede the onset of cognitive decline in Alzheimer's disease patients and then progress in parallel with both cognitive dysfunction and the progression of Alzheimer's pathology. This is a vicious cycle as they progress in tandem.
Using pet scans, researchers have identified those reporting greater than seven hours of sleep have the lowest amyloid burden. Those that have between six and seven hours have an intermediate amyloid burden. Those with less than, or just six hours of sleep, have the most significant amyloid burden. Again, this is in older adulthood, and the studies have identified that some of these changes begin to occur around age 50. Recall that the mechanisms of Alzheimer's are already at play before the first signs and symptoms are present. So, if we see those signs and symptoms around age 60, we need to address sleep needs in midlife.
Moreover, those who have preclinical amyloid pathology, meaning not yet present, have increased napping. Napping might be an indication of disordered sleep. We know that older adults frequently nap throughout the day, which causes disrupted sleep at night. Additionally, naps do not seem to have the same clean-out quality as organized rest later in the evening.
- Sleep Factors
- Normal Aging
- ↓ in non-REM short wave sleep
- ↑ in lighter sleep stages
- ↑ in sleep fragmentation
- Normal Aging
- ↑ Alzheimer’s Risk
- Sleep-disordered breathing or untreated sleep apneas
- Even greater fragmentation
- Shorter overall sleep duration
- Lower sleep quality
- Leading to excessive daytime sleepiness and napping
We also know some of these factors, like decreasing non-REM short wave sleep in normal aging. Older adults have lighter sleep stages, meaning they are more likely to be awoken from sleep. Thus, they have more significant sleep fragmentation and have a more challenging time getting that consolidated X number of hours of sleep. So, the very thing we need is the most difficult to get as we age.
What are the differences in those at higher risk of Alzheimer's disease? There are a few factors. It tends to be a matter of degrees. Those with the most significant dysfunction tend to be at the highest risk. Sleep-disordered breathing or untreated sleep apneas are substantial because this can affect the amount of oxygen that the brain receives. Individuals with greater sleep fragmentation and many nighttime awakenings (those that are getting up and walking around during the night or engaging in activities that do not promote a return to sleep) are those older adults that our education and sleep hygiene can benefit. Those with the highest degree of Alzheimer's risk have shorter overall sleep duration and the number of sleep hours in the evening. And, their sleep quality is poor, which causes excessive daytime sleepiness and napping during daytime hours.
Protective Factor Programming
Given all of that, what are some of the programmatic things we can do to address some of these protective factors in our older adult populations?
Awe Walk
- Targets perspective-taking and pro-social emotions during a walking activity
The awe walk is exciting to me. This work came out of the state of California with researchers from UC Berkeley and San Francisco State. They were targeting perspective-taking and pro-social emotions brought on by the experience of awe during a walking activity. The hypothesis is that feelings of awe tend to diminish our self-importance and make us feel smaller and more connected. These feelings may help to elevate our sense of compassion, admiration, gratitude, engagement or connection, and empathy for others. These things are called pro-social emotions.
- “a positive emotion…in the presence of something vast that (we) cannot immediately understand”
- Nature, art, music, collective action, courage…
- ↓ anxiety, depression, cortisol, inflammation, cardiovascular risk factors
- Can be nurtured and cultivated
The researchers identified awe as a positive emotion in the presence of something vast that we cannot immediately understand. Awe can be found in various places, like nature, art, music, collective action, or significant acts of courage, and is very individualized. In this awe walk, they were looking for a decrease in anxiety, depression, and lower cortisol levels.
We just talked about stress hormones, lower levels of inflammation, and cardiovascular risk factors that affect overall health. Researchers proposed that a sense of awe can be nurtured and cultivated in daily activity.
- Group Design
- "Control Walk"
- 28 participants
- At least one 15 min outdoor walk weekly for 8 weeks
- No additional instructions
- "Awe Walk"
- 24 participants
- At least one 15 min outdoor walk weekly for 8 weeks
- Received additional instructions (provided in handouts)
- "Control Walk"
Their design had 28 participants in a control walking group and 24 participants in an awe walking group, which yielded an 80% power analysis using a two-sample T-test. Volunteers were recruited from the community, and these were older adults between the ages of 60 and 90 that were participating in the healthy aging network at the UCSF system. They were randomly assigned to one of these two groups and completed at least one 15-minute outdoor walk weekly for eight weeks. In the control walk group, they received no other instructions, but in the awe walk group, they received additional instructions.
- “With the right outlook, Awe can be found almost anywhere”
- Must be outside
- Minimize phone usage, except for pictures
- 3 pictures of yourself (before, during, and after) and 1 of something else (the most interesting thing you saw)
The awe walks had to be outside and not in a built environment. Although we may find awe in a particular architectural structure, as an example, "awe" is more commonly accessible to be outside. They requested that walkers have minimal phone use on their walk other than for pictures that they were asked to take. The subjects in both groups took three pictures of themselves, one before the activity, one during the activity, and one after the activity. They were also instructed to take another picture of the most interesting thing they saw on their walk. The walks were specified to be alone, but participants in both groups took along a companion in equal measure between the control group and the awe walk group.
- Results
- "Control Walk"
- More frequent walks (18.9)
- No statistically significant difference in speed, duration, or distance of the walks between groups
- No change in self-size perception
- "Awe Walk:
- ↑ Increases in:
- emotions of ‘awe’, compassion, admiration, appreciation, joy, pride
- pro-social positive emotions
- smile intensity over time
- ↓ Decreases in:
- self-size perception
- distress over time
- a daily sense of sadness and fear (but not anger, anxiety, or annoyance)
- ↑ Increases in:
- "Control Walk"
Significantly and surprisingly to the researchers, the control group walked more than twice as often as the study group. The participant's expectation was at least one 15-minute walk per week over the eight weeks of the study. The awe walk group completed the minimum number of walks assigned; whereas, the control group completed more than twice that with about 18.9 walks over the eight weeks. All participants were blinded to the group assignment. It was hypothesized that without additional instructions as to the walk's purpose, the control group may have assumed that their fitness or their frequency of walking was being tested. And as a result, the control group spent more time walking.
In contrast, the awe walk group had specific instructions and a specific task, so they did not perform more than required for their participation. They had a job to do, and they fulfilled it. What was also noted in the two groups is that there was no significant difference in speed, duration, or distance between the two groups. They both walked about the same rate, for the same amount of time, and the same length.
The researchers also looked at the images they took. They looked at the number of pixels devoted to the individuals in their selfie versus the background. Remember, they were taking pictures of themselves on the walk. They proposed that the individuals, in the presence of awe, would be smaller as the self is diminished. They then correlated this with questionnaire responses that reported greater increases with feeling a part of something larger than themselves during the awe walk than the control group.
The results showed that neither group reported feeling that they were in the presence of something vast, which might reflect the environment more than the personal perspective. It can be challenging for us, in some environments, to be exposed to something we would call vast. In the awe walk group, when looking at pro-social positive emotions (like awe, compassion, admiration, appreciation, joy, or pride), researchers did observe an increase in the research groups' smile intensity over time. Meaning in their selfies, the awe walk group tended to have more significant smiles and decreases in their self size perception. As they reported being in the presence of awe, their size in the selfie grew smaller. They also reported declines in distress and a daily sense of sadness and fear, but not anger, anxiety, or annoyance. If we think about that, this is not necessarily surprising because anger, anxiety, annoyance tend to be very episodic feelings. We can be annoyed about a particular thing versus a sense of sadness, and fear may be more chronic.
What was also interesting is that the effects were not visible in one single walk event. When comparing one control walk to one awe walk, the results were relatively small. Thus, a single event was not necessarily correlative but was significant relative to time. The difference was not a one-time event, but if the elder practiced it consistently over time, they showed greater progressive gains in all measured areas. Researchers called that a dose-like relationship, meaning the more you do it, the more benefits you get. Those of us that have mindfulness practices would say that it occurs like a practice. Once you have a practice of looking for awe, then you have more access to these benefits.
The Balanced Life Group
- “To inspire and promote balance in overall wellness”--Zechner & Kirchner
Another protective factor program present in the literature is called the balanced life group. This group was for older adults who were hospitalized for psychiatric disorders. Due to its protective factors, I think it is worth reexamining here in this context as well. A multidisciplinary team developed this group to inspire and promote balance in overall wellness. As a programmatic tool, this has a lot of benefits for our older adults.
- Group Design
- 9 topic areas
- 45-min sessions
- 2x weekly over 11 weeks
- Application of knowledge and personal experiences
- A strengths-based approach, open-ended questions
- 2 staff co-facilitators provided links between topics and summarized the learning
- Each topic’s first session = introduced new material
- Second session = facilitation to personalize the material
- Final summary = complete a wellness plan, describe strengths, identify one area to improve in each domain
There were nine topic areas and 45-minute sessions that occurred twice weekly over 11 weeks. The group focused on a strengths-based approach with two co-facilitators that provided the links between the topics. Each topic's first session introduced new material relative to that topic; whereas, the second session facilitated the personalization of that material by asking questions like, "How is that relevant to you?" Then, there was a final summary of the group overall with each individual's completed a wellness plan that described their strengths and one area to improve in each domain.
- Group Modules
- Spirituality (2 sessions)
- Physical (2 sessions)
- Metabolic Syndrome (1 session)
- Emotional (2 sessions)
- Intellectual (2 sessions)
- Social (2 sessions)
- Occupational (2 sessions)
- Environmental (2 sessions)
- Sexuality (1 session)
Above are the group modules.
- Sample Final Topic Summary
- My Balanced Life Plan
- Spiritual Wellness:
- My strengths in spirituality are: ___________________
- One thing I would like to do or continue to do: ______
- Physical Wellness:
- My physical strengths are: ________________________
- One thing I would like to do or continue to do: ______
- …continued
- (metabolic syndrome and sexuality [one session] topics were informative/educational and omitted from goal setting)
In their final summary, they developed a balanced life plan.
- Participant Feedback
- “This group reminded us that we are still human beings”
- “Good to have perspectives on different areas in life”
- “Look at the whole picture of your life and try to improve”
- “Talking about our past experiences and how they contribute to wellness was different than other groups”
- “I would like to see a manual so that I would have the stuff in front of me in case I forgot about what was covered in sessions”
The feedback on these topics was fascinating because participants liked that they were reminded of the importance of balance in their lives and to look at the whole picture of your life and try to improve. I think we see, throughout the literature, the importance of resilience and the ability to have a locus of control over your life despite your surroundings or experiences.
Strengths vs. Aerobics for Residents with Dementia
- Comparing strength training to aerobic training and measuring its impact on ADLs, cognition, depression, and biomarkers
Let's now talk about some of the findings relative to exercise in particular. This is one of the studies I mentioned that focus on the value of strength versus aerobics for residents with dementia. They measured the impact of exercise on ADLs, cognition, depression, and various biomarkers. This was published mid-pandemic in 2020 by the American Congress of Rehabilitation Medicine.
- Participants
- 69 initiated (- 8 for intolerance)
- Residential skilled nursing environment (veterans)
- 50 men, 11 women
- Ages 66-95 (85.7 avg.) with MMSE between 15-26
- (able to complete an informed consent and answer questionnaire questions)
- Excluded for disorders affecting safe participation
- Randomly assigned to the strength training group or aerobic training group
Sixty-eight participants started in a residential skilled nursing environment, but they lost eight that did not tolerate the exercise. It was also specifically for veterans, which explains some of the gender differentials there. They had a very clinical age group, which is good to see. Sometimes studies are performed on younger individuals when our clinical populations are much older. The average age of the participants was just over 85. The mini-mental status exam scores were between 15 and 26 because they needed to provide informed consent. Thus, their levels of dementia were relatively low. Participants were also excluded for any disorders that might affect their safe participation and were randomly assigned to either a strength or an aerobic training group.
- 4-Week Programs
- Strength Training (n=30)
- isotonic weight training machines targeting the specific muscle groups on alternating days:
- biceps, triceps, pecs major
- glutes and quads
- 40-50% of 1 rep maximum
- 2 sets of 12 reps daily
- 5 days/week
- Strength Training (n=30)
- Aerobic Training (n=31)
- stationary bicycle
- pedaling at PRE level of 5-6 on a 0-10 scale
- 30-min sessions
- 5 days/week
This was a four-week program with either weight training machines five days a week up to two sets of 12 reps, or a stationary bicycle for the aerobic group with a perceived rate of exertion between five and six on a 10-point scale, 30 minutes, five days a week.
- Results after 1 mo.
- Strength Training
- ADLs (Barthel)
- ↑ cognition using MMSE
- ↑ cognition using MoCA
- ↓ inflammation (MCP-1 )
- Aerobic Training
- ↑ ADLs (Barthel)
- ↑ cognition using MMSE
- ↑ cognition using MoCA
- ↑ upregulation of BDNF
- ↓ inflammation (MCP-1 )
- Strength Training
After one month, both groups had an increase in their ADL performance on the Barthel index. They also had an increase in their cognition using a mini-mental status exam as well as the MoCA. There was a decrease in inflammation as measured by the MCP-1. Only the aerobic training group had an increase in the upregulation of BDNF, a brain-derived neurotrophic factor. By and large, the benefits were observed in both the strength training and the aerobic training fitness groups.
Seated Fitness with Resistance Bands
- Seated exercise ensures the distance between participants and minimizes cleaning protocols while providing an evidence-based ‘low-load’ improvement in functional fitness.
This seated fitness with resistance bands article was released in 2021. The target audience was older women. This study was particularly exciting because it was during the pandemic. It ensured appropriate distance between participants as they were sitting in chairs with their resistance bands. This might be a great group approach.
- Group Participants
- 6 participating institutions
- Elder women in residential facilities
- Excluded for disorders affecting safe participation
- No organized physical activity in the prior 2 years
- The study group (n=90) followed a structured resistance training
- Control group (n=90) engaged in usual residential programming (reading, crafts, games, etc.)
Again, this focused on elderly women in six different residential facilities, both long-term and skilled nursing environments. Excluding those with orthopedic, neurologic, cardiovascular disorders that might affect their participation, most had no specific physical activity in the prior two years, typical for this population. The study group had 90 participants that followed a structured resistance band training along with a control group. The control group still participated in the usual programming available to the residents, such as crafts, games, things in the activity room, et cetera.
- Group Design
- 12-weeks duration
- 10 residents per group
- 2 co-instructors
- Light intensity t-band
- 1.5m relaxed length
- 2 sets of each activity
- 12-15 reps in each set
- 3 seconds cadence
- 1 min rest
- RPE: 4-5
- 12 chair-based exercise band activities
- knee flexion
- knee extension
- bent row
- chest press
- hip flexion
- obliques
- upright row
- trunk flexion
- biceps arm curl
- overhead triceps extension
- hip abduction
- standing hip extension
Above is the specific group design.
- T-band Program Outcomes
- High adherence
- 22+/- 1 of 24 sessions (92%)
- No reported side effects
- ↑ Tx & Control
- 30-sec chair stand
- 30-sec arm curl
- 2 min step test
- ↑ Treatment Group
- Chair sit and reach
- ‘Back scratch’
- 8 ft Up and Go
- Hand grip
The participants had high adherence, with 92% completing all sessions with no reported side effects. Interestingly, we know that some activity is better than no activity. Thus, in both treatment and control groups, those in other seated activities showed improvement in the 30-sec chair stand, 30-second arm curl, and 2-minute step test. The benefits of general activity versus the benefits of the resistance band group also showed increases in their chair sit and reach flexibility, their back scratch test, 8 Ft Up-and-Go, and their hand grip strength.
Sensory Memory Workshop for Dementia
- Paired cognitive and sensory stimuli may improve engagement and reactivate memory and connection post-isolation.
Here is a specific memory workshop. Recall that the effects of the environment are also good resilience factors for neural network efficiencies. Here are paired cognitive and sensory stimuli focusing on a group design to reactivate memory and connection following a period of isolation.
- Group Design
- Small group of men with dementia (n=5)
- Mild or moderate severity
- 7 meetings per set
- Based on sensory organs and cognitive stimulation
- Coordinated by occupational therapy
This was a small group of men (n=5) with dementia. While it was a small sample size, it had an excellent plan design. There were seven meetings in the group, and each one focused on a different sensory system, as well as cognitive stimulation threaded throughout. This was coordinated and led by occupational therapy.
- Session Plans: 1-3
- 1st Session: Orientation & Greeting/Meeting
- 2nd Session: Planning
- Explain objectives
- Discuss personal interests
- Encouragement to expand choices, preferences, desires, and memories
- 3rd Session: Smell
- 9 types of food with strong aromas (lemon, oregano, ground parmesan, orange, banana, vinegar, mint, fennel, popcorn) hidden in a closed cup
- Participants matched smell to a large laminated picture
The first session focused on orientation, the second session assessed personal interests, and the third session focused on smell. Items with strong aromas were hidden in a closed cup. Participants were asked to match the smell to a large laminated picture. What is exciting about this model is that you can make some cultural adjustments based on your population. You can incorporate some culture-specific foods and aromas in this particular design.
- Session Plans: 4-5
- 4th Session: Tasting
- Fruit Salad Activity in Two Parts
- Part 1: Recall of fruits used in fruit salads and identifying by their touch or smell (not vision)
- Part 2: Preparation of the fruit salad: washing peeling, and cutting fruit, mixing, & serving
- 5th Session: Hearing
- Listening to music (from videos) reflecting preferences collected in Session 2.
- Part 1: Listening to audio only (no video) for 3 minutes each and recalling memories from the music
- Part 2: Listening again while watching the video for visual
- 4th Session: Tasting
The fourth session was a two-part activity of preparing a fruit salad. First, they talked about fruit salad by identifying different fruits by touch or smell and not using their vision. In the second part of the activity, they prepared the fruit salad by washing, peeling, cutting, and mixing it before serving it. The fifth session was designed around hearing. They listened to music from videos that reflected their preferences. They listened to the audio-only for three minutes and stimulated their recall from the music on their own. In the second part, it was paired with the visual stimulus of the music video. This was to create some new neural pathways to increase their resilience and connection.
- Session Plans: 6-7
- 6th Session: Conversation Circle
- Discuss perception of the experience
- How senses support our independence and autonomy
- 7th Session: Thematic Party
- Fostering connection and closing of the cycle
- 6th Session: Conversation Circle
The final sessions included a conversation circle and thematic party to close off the cycle of activities.
Summary
- Top 5 Protective Factors
- Cognitive Reserve
- Physical Activity/Fitness
- Environment
- Socialization
- Sleep
To summarize, there are the top five protective factors, which we discussed today. These are cognitive reserve, physical activity and fitness, the environment, socialization, and the importance of sleep.
Questions and Answers
Is there evidence to suggest that taking on new skills or tasks in the early stages of disease could reduce the disease process?
I think this is an interesting question because it goes into cognitive reserve and neural efficiency. The researchers are addressing years of education because higher learning requires us to learn, think, and process information in new ways. Once we stop doing new learning, we tend to form the same cognitive pathways over and over. Learning a new activity requires us to create a different neural pathway. One thing also related to the issue of resilience is that the brain, if it has that opportunity, will form new connections and access the same information via a different route due to brain plasticity.
Would diet be an environmental aspect?
There is a lot of literature about diet and its effect on overall health, specifically diabetes and cardiac issues. I did not include diet in the top five protective factors because the connection specifically to cognitive health is still emerging. As with physical activity and fitness, we know that diet has some benefits, but we do not know how that occurs. If you make changes in diet and any other health improvements that reduce your incidence of, say diabetes, we know this will improve overall health. However, the studies that directly tie it into cognitive health are still emerging.
Is there any research suggesting women on aromatase inhibitors for breast cancer are at increased risk of dementia due to sleep disturbance and brain fog?
This is a great question. And again, this does not have a direct connection, but the decreased sleep is significant. We can take a few steps in our clinical reasoning there and say that anything that affects sleep could potentially be a factor in cognitive health. One finding from sleep research is that a C-PAP introduced early in the Alzheimer's disease process can help with sleep and the ability of the brain to clean out the proteins. Sleep-disordered breathing tends to be underdiagnosed in older women, and we tend to see sleep apnea more commonly diagnosed in men. It is not a surprise that sleep apnea is underdiagnosed in women, yet women have a higher incidence of Alzheimer's disease. Both of those are related factors.
Thank you for your time today. And if you have any further questions, I will refer you to the references. Again, particularly that study on sleep. There are several interesting findings, both for your life and your sleep practice, working with adults and older adults. Thank you so much.
References
Babulal, G.M. Quiroz, Y.T., Albensi, B.C., Arenaza-Urquijo, E., Astell, A.J., Babiloni, C., Bahar-Fuchs, A., Bell, J., Bowman, G.L., Brickman, A.M., Chételat, G., Ciro, C., Cohen, A.D., Dilworth-Anderson, P., Dodge, H.H., Dreux, S., Edland, S., Esbensen, A., Evered, L., Ewers, M. et al. (2019). Perspectives on ethnic and racial disparities in Alzheimer's disease and related dementias: Update and areas of immediate need. Alzheimer's & Dementia, 12(2), 292-312. https://doi.org/10.1016/j.jalz.2018.09.009.
Biogen. (2021). Amyloid-beta cascade hypothesis. Identify Alzheimer’s Disease Earlier. https://www.identifyalz.com/en_us/home/pathophysiology/amyloid-beta-cascade-hypothesis-alzheimers-disease.html
Center for Disease Control and Prevention (2020, October 26). Alzheimer’s disease and related dementias. Alzheimer’s Disease and Healthy Aging. https://www.cdc.gov/aging/aginginfo/alzheimers.htm
Center for Disease Control and Prevention (2018, September 20). U.S. burden of Alzheimer’s disease, related dementias to double by 2060. CDC Newsroom. https://www.cdc.gov/media/releases/2018/p0920-alzheimers-burden-double-2060.html
Dumurgier, J., Paquet, C., Benisty, S., Kiffel, C., Lidy, C., Mouton-Liger, F., Chabriat, H., Laplanche, J-H., & Hugon, J. (2010). Inverse association between CSF Aβ 42 levels and years of education in mild form of Alzheimer's disease: The cognitive reserve theory. Neurobiology of Disease, 40(2), 456-459. https://doi.org/10.1016/j.nbd.2010.07.007
Liu, I.-T., Lee, W.-J., Lin, S.-Y., Chang, S.-T., Kao, C.-L., & Cheng, Y.-Y. (2020). Therapeutic effects of exercise training on elderly patients with dementia: A randomized controlled trial. Archives of Physical Medicine and Rehabilitation, 101(5), 762–769. https://doi.org/10.1016/j.apmr.2020.01.012
Mander, B. A. (2020). Local sleep and Alzheimer’s disease pathophysiology. Frontiers in Neuroscience, 14, 525970. https://doi.org/10.3389/fnins.2020.525970
Martins, R. N., & Brennan, C. S. (2019). Neurodegeneration and Alzheimer’s disease : The role of diabetes, genetics, hormones, and lifestyle. John Wiley. https://doi.org/10.1002/9781119356752
Silva, M. V. F., Loures, C. de M. G., Alves, L. C. V., de Souza, L. C., Borges, K. B. G., & Carvalho, M. das G. (2019). Alzheimer’s disease: Risk factors and potentially protective measures. Journal of Biomedical Science, 26(1), 33–33. https://doi.org/10.1186/s12929-019-0524-y
Citation
Fair-Field, T. (2021). The role of resilience in Alzheimer's dementia. OccupationalTherapy.com, Article 5466. Retrieved from http://OccupationalTherapy.com
