Introduction
Clear objectives and explicit communication are important to the success of any program, and even more so in these times of rapid and dramatic change. As occupational therapists, we strive to create meaningful change in our patients’ lives utilizing services that are skilled and reimbursable, while also ensuring utmost efficiency and productivity. This is no easy task; it requires continual assessment of our outcomes, which is impossible if our methods are vague or ambiguous. Unless a clinician can accurately identify and measure the changes associated with treatment, it is difficult to ensure good use of resources and reproducibility of results. It is also important that our services are considered necessary and effective for reimbursement purposes. It is therefore imperative that our evaluations and treatment techniques are clear, measurable, and easily interpretable for insurance carriers.
Effective communication between patient and therapist is also crucial for achieving satisfactory outcomes. Do our patients truly understand the treatment process? Are we providing the patient with sufficient feedback to encourage involvement or do our methods encourage dependency on the therapist? Patient engagement is important for retention of gains made during therapy. It ensures that the patient is not a subject, but a partner, one who is involved in every step of the healing process.
Frequent organizational changes and high turnover of staff, common features of most rehabilitation settings, lead to complex changes for everyone involved. In these situations, use of subjective methods that cannot be clearly communicated among clinicians can be demoralizing and a hindrance in the path to achieving treatment goals.
Utilizing practices and tools that provide the patient with adequate feedback and can be clearly communicated among clinicians and insurance carriers is therefore essential. One such tool is biofeedback, a widely recognized mind and body practice under complementary health approaches.
Background
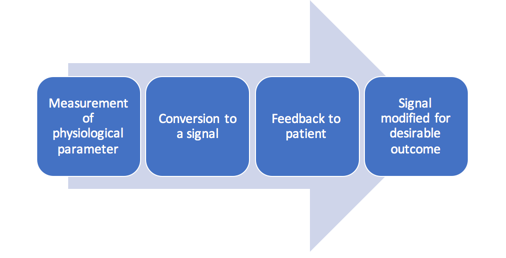
Several streams from various sources, including relaxation therapy, autogenic training, diagnostic EMG, single motor neuron training, operant conditioning, and electroencephalography (EEG), came together in the 1960s to form the current practice of biofeedback. Dr. John Basmajian, known as the “Father of EMG Biofeedback”, isolated motor units and activated them consciously using fine-wire electrodes. His work on single motor unit training provided the momentum for biofeedback research (Basmajian, 1989; Schwartz, 1995).
Types of Biofeedback
Some of the common types of biofeedback are:
- EMG: measures electrical activity, a correlate of muscle contraction
- Real-time ultrasound imaging: uses sound waves that are reflected by body tissues and displayed on a screen, providing real-time feedback of tissue activity
- Biomechanical: measures movement, postural control and forces produced by the body
- Peripheral temperature: measures temperature changes caused by vasodilatation/vasoconstriction
- Electrodermal: measures the galvanic skin response or skin conductance, which corresponds to sweat gland activity
- Photoplethysmography: measures peripheral blood flow, heart rate, and heart rate variability
- EEG/Neurofeedback: measures brain wave activity
- Pneumography: measures respiration rate and relative contraction/expansion of chest and abdomen
- Capnography: measures end-tidal CO2
- Homoencephalography: measures differences in light color reflected through the scalp based on the amount of oxygenated and unoxygenated blood in the brain (Lockheed, 2010).
This article describes the use of EMG biofeedback and its applications in rehabilitation based on current evidence.
Surface EMG (SEMG)
SEMG is a safe, non-invasive tool that provides an objective display of neuromuscular activity, which can be used to assess the success of treatment techniques and assist in further enhancement of appropriate strategies. Feedback provided by SEMG improves patient learning and confidence in the process. It also improves communication among patients, clinicians, and insurance carriers.
Instrumentation
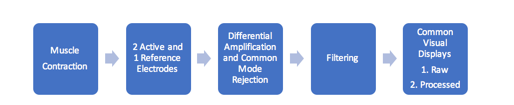
The source of the SEMG signal is the motor unit action potential. During a muscle contraction, motor units are activated in an asynchronous pattern and give off action potentials. This energy reaches the skin, is sensed by the electrode, and passes through the process of differential amplification and common mode rejection. It is then filtered to reduce artifacts and is finally displayed as a visual signal or an auditory tone (Basmajian, 1989; Cram, Kasman, & Holtz, 2005) (Figure 2).
SEMG terminology
- Impedance: Skin impedance refers to the resistance (ohms) at the electrode-skin interface. It is related to the moisture level of the skin and the density of the dead-cell layer. This impedance should be lower than the input impedance of the SEMG preamplifier.
- Common Mode Rejection and Differential Amplification: Biological energy common to 2 recording electrodes, referred to as the common mode, is rejected, and the remainder is amplified.
- Filtering: This refers to processing of the signal to eliminate electrical noise from the recording environment.
- Artifact: A source of noise that is mixed with the SEMG signal and needs to be reduced/eliminated is termed an artifact. Biological artifacts include:
- ECG- picked up on torso sites, especially on left side of the body
- Respiration- picked up on upper torso and neck sites
- Cross-talk- energy of other muscles reaching the recording electrodes
- Other artifacts include:
Movement artifact: This is caused by movement of the electrode on the surface of the skin.
60-cycle artifact: This refers to surrounding sources of 60-cycle energy such as computers, lights etc.
Radio frequency: This is caused by local radio signals (Basmajian, 1989; Cram et al., 2005; Schwartz, 1995).
Static and Dynamic EMG assessment
Types of SEMG displays
- Visual displays: The 2 most common forms of visual display are a) Raw SEMG- This is the unprocessed signal that includes all the artifacts and the positive and negative directions, which makes it difficult to interpret during biofeedback training (Figure 3); b) Processed SEMG- Here, the signal is rectified, that is the entire signal is made positive and placed above the crossing line (Figure 4) and then smoothed using integral averaging.
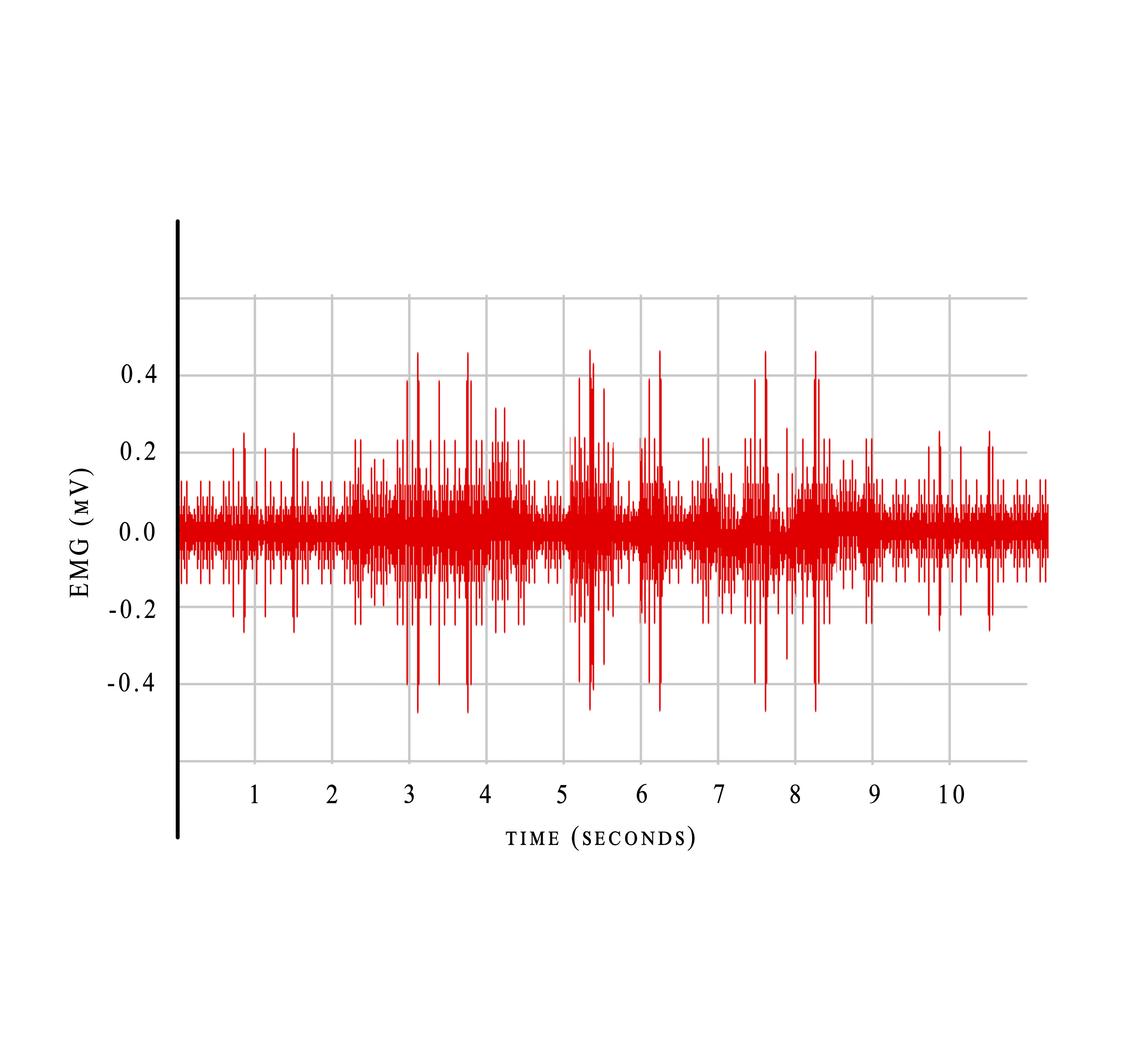
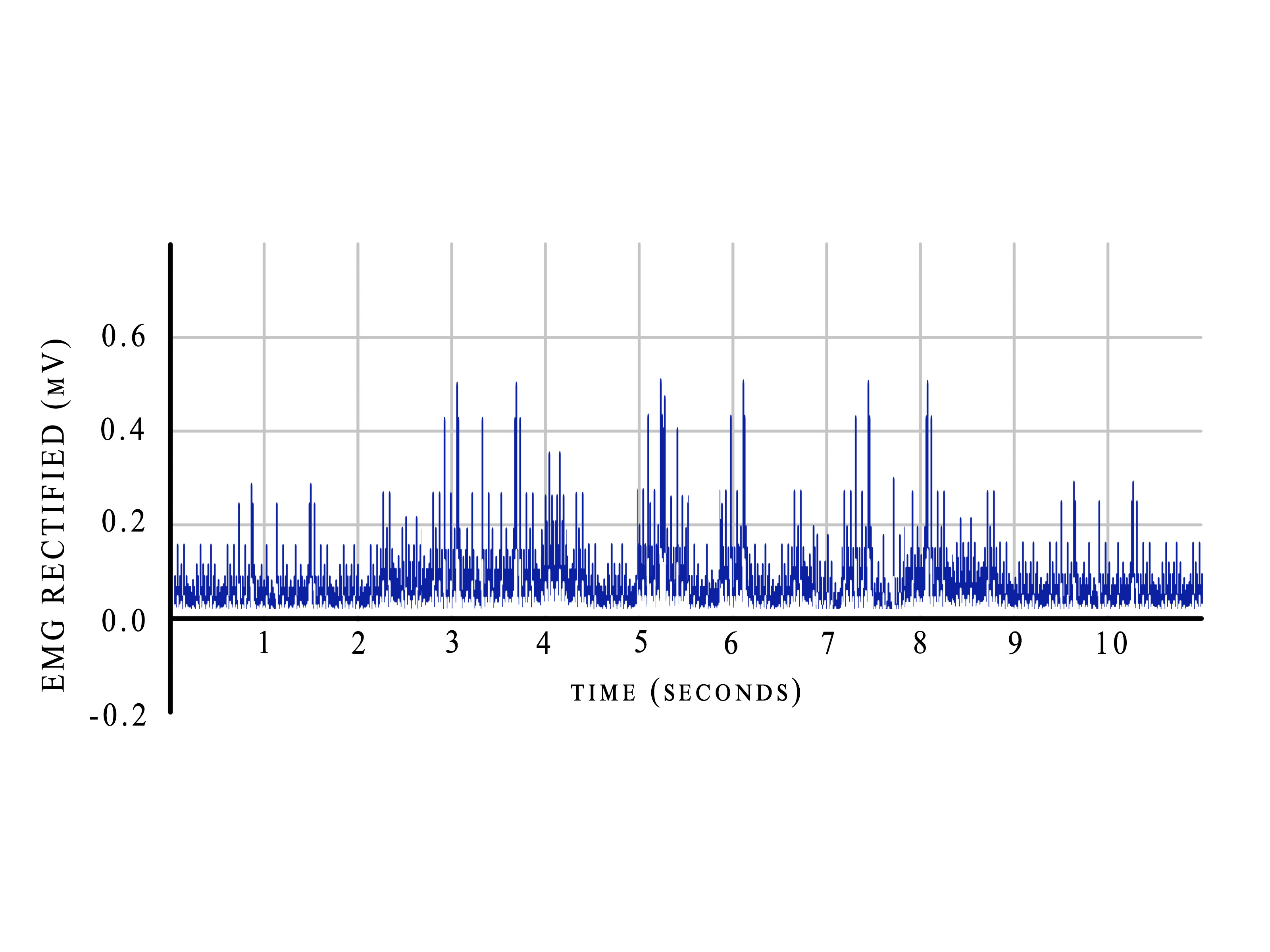
Figure 4. Rectified EMG.
- Auditory displays: These can be in the form of a continuous tone or a pulsed tone, where a high pitch indicates a high amplitude (Basmajian, 1989; Cram et al., 2005; Schwartz, 1995).
Biofeedback Devices
There is a wide variety of biofeedback devices available, both stand-alone and computerized systems, ranging from a few hundred to thousands of dollars in price. Several factors need to be taken into consideration when selecting the correct biofeedback device. These are:
- FDA approval
- type(s) of intervention(s) provided
- number of channels required and displayed simultaneously
- portability
- user-friendliness of software and hardware
- cost of other accessories such as pre-gelled electrodes, sensor pads, etc.
Figure 5 shows an 8-channel system, with 2 channels for viewing raw EMG, EEG, and EKG signals. The remaining 6 channels can be used with any combination of sensors, including EEG, EKG, RMS EMG, skin conductance, heart rate, blood volume pulse, respiration, goniometry, force, and voltage input. It can capture data in real time by connecting directly to the PC via fiber-optic cable, or it can store data on a Compact Flash memory card for uploading later to the PC.
Figure 6 shows a 2-channel stand-alone SEMG monitor that allows measuring and monitoring 2 channels of high-resolution SEMG in a large number of configurations.
Figure 7 shows pre-gelled single-use disposable SEMG/ECG electrodes, ideal for placement on dry skin.
Figure 8 shows silver/silver chloride (Ag/AgCl) - electrode pellets (approx. 8 mm in size) for SEMG or EEG and for use with any standard snap cable (Bio-medical instruments, 2017).

Figure 5. ProComp Infiniti. Image used with permission of Thought Technology.

Figure 6. MyoTrac Infiniti 2. Channel SEMG 60 Hz. Image used with permission of Thought Technology.

Figure 7. Uni-Gel electrodes. Image used with permission of Bio-Medical Instruments Inc.

Figure 8. EMG/EEG electrode pellets. Image used with the permission of Bio-Medical Instruments Inc.
It is important that the clinician is knowledgeable of the electronic specifications of the various EMG devices available. A discussion of these specifications is beyond the scope of this article, but is available in detail in most books on biofeedback (Basmajian, 1989; Cram et al. 2005; Schwartz, 1995).
Electrode Placement
An important limitation of SEMG lies in its specificity secondary to “cross-talk”, where the recording may be mixed with energy from the surrounding muscles. Selection of the most specific electrode sites helps diminish the cross-talk and isolate the recording to the target muscle group. Clinicians can learn more about these specific sites from available guides (Basmajian, 1989; Cram et al., 2005).
Important considerations for electrode placement
- Size of electrodes and interelectrode spacing: depends on the size and area of the muscles being recorded
- Adhesion of the electrodes: depends on the muscle and type of recording, with strong adhesion essential in cases of muscle stretching and movement
- Site preparation: rubbing the electrode site using an alcohol pad, use of pre-gelled electrodes/application of gel on recording site to decrease impedance at electrode-skin interface
- Preferred placement for maximum sensitivity: parallel to muscle fibers being recorded
- Site selection: to minimize cross-talk and avoid problematic areas that would make recording or movement difficult
Clinical Applications of EMG Biofeedback
There is a large body of evidence for EMG biofeedback in rehabilitation. It has been found to be effective in several musculoskeletal and neurological conditions. This section discusses current evidence for various applications of EMG biofeedback.
Incontinence
Biofeedback has been found to be effective in dysfunctional voiding in children. Dysfunctional voiding is characterized by urinary incontinence, recurrent urinary tract infections (UTI), and nocturnal enuresis in children. It is associated with dyssynergic sphincter activity that is unrelated to neurological or structural etiology. In a recent study of 45 children (34 girls, 11 boys; age range 5-15 years) with dysfunctional voiding, the patients were first treated with standard urotherapy, and the changes in symptoms were recorded. They were then treated with 6-10 sessions of biofeedback, which involved use of a video game that makes patients move objects on the screen by flexing their pelvic floor muscles. EMG electrodes were placed on both sides of the perineum and on the rectus muscle to measure abdominal muscle activity. Patients were also given home exercises. Combined urotherapy and biofeedback treatment was found to be significantly more effective than standard urotherapy alone in decreasing urinary incontinence, UTI occurrence, and post-void residual urine volume; it was also found to be more effective in correcting voiding patterns (Altunkol et al., 2018).
A randomized controlled trial (RCT) conducted in 2017 found a combination of EMG biofeedback and pelvic floor exercises to be more effective than pelvic floor exercises alone in improving muscle strength and pre-contraction in postmenopausal women with stress urinary incontinence (Bertotto, Schvartzman, Uchôa, & Wender, 2017).
EMG biofeedback-assisted pelvic floor muscle therapy was also found to be effective in decreasing symptoms of the overactive bladder syndrome in women in another RCT (Voorham et al., 2016).
Pain
A pilot study with a small sample size of 15 manual wheelchair users with spinal cord injury provided preliminary evidence for the benefits of adding EMG biofeedback to a 10-week exercise program for reducing shoulder pain (Middaugh, Thomas, Smith, Mcfall, & Klingmueller, 2013).
SEMG-assisted biofeedback stretching has also been shown to improve the flexion-relaxation response, which is impaired in individuals with chronic low back pain (Moore, Mannion, & Moran, 2015; Neblett, Mayer, Brede, & Gatchel, 2010).
A recent meta-analysis and systematic review of RCTs assessed the efficacy of EMG- and EEG-biofeedback in fibromyalgia syndrome. It included 7 studies (321 patients) on EEG biofeedback and EMG biofeedback. The mean age range of the participants was 32–57 years. The participants were predominantly female and white. The number of sessions varied between 6 and 22 sessions and the length of sessions varied between 30 minutes and 3 hours. The analysis revealed that only EMG biofeedback and not EEG biofeedback significantly reduced (short-term) pain intensity in comparison to that seen in the control group.
However, the long-term effects of EMG biofeedback on pain intensity are unclear due to the small number of studies that included follow-up assessments. Additionally, biofeedback did not reduce sleep problems, depression, fatigue, or health-related quality of life (QOL) in comparison to the control group (Glombiewski, Bernardy, & Häuser, 2013).
Stroke
In a recent study of 30 patients with stroke of disease duration longer than 6 months, those receiving intensive EMG biofeedback showed more significant upper extremity functional recovery than those who received only traditional rehabilitation therapy (Kim, 2017).
In another study of 16 patients with ischemic stroke, those who participated in myofeedback training along with physical therapy demonstrated greater improvement in spasticity levels compared to those who participated in traditional physical therapy rehabilitation (Vieira, Silva, Melo, & Soares, 2016).
Kim, Cho, Kim, and Lee (2016) found that ankle biofeedback training significantly improved strength, balance, and gait compared to ankle strengthening exercises alone in patients with stroke.
Lourenção, Battistella, Brito, Tsukimoto, and Miyazaki (2008), in their study of 59 patients with stroke, concluded that those receiving EMG biofeedback demonstrated significantly greater improvement in range of motion and recovery of upper extremity function than those receiving only occupational therapy and functional electrical stimulation.
However, in a preliminary clinical trial conducted on 30 patients with stroke, neurofeedback and EMG biofeedback therapy were not found to be superior than conventional occupational therapy for improving hand function in stroke patients (Rayegani et al., 2014).
Cerebral palsy
Prolonged electromyographic biofeedback has been found to improve upper extremity function in children with cerebral palsy (Bloom, Przekop, & Sanger, 2010).
EMG biofeedback when augmented by virtual reality exercise games produced better neuromuscular balance control in the elbow joint than EMG biofeedback alone in a study on children with spastic cerebral palsy (Yoo, Lee, Cha, & You, 2017).
Shoulder
A common exercise used in rotator cuff retraining programs is shoulder external rotation while maintaining adduction to increase stabilization and strength of the infraspinatus. Shoulder adduction isolates the infraspinatus from the posterior deltoid, and the side-lying external rotation exercise may be the most effective maneuver to increase infraspinatus activation.
The effect of visual EMG biofeedback during side-lying shoulder external rotation exercise on posterior deltoid and infraspinatus activation and the infraspinatus/posterior deltoid EMG activity ratio was evaluated by Lim, Kim, Song, Cynn, & Yi (2014). Thirty-one asymptomatic subjects were included. Subjects performed side-lying shoulder external rotation exercise with and without visual EMG biofeedback. SEMG was used to collect data from the posterior deltoid and infraspinatus muscles. Visual EMG biofeedback applied a pre-established threshold to prevent excessive posterior deltoid muscle contraction. Significantly lower posterior deltoid activity was noted, while infraspinatus activity and the infraspinatus/posterior activity ratio significantly increased during the shoulder external rotation exercise with visual EMG biofeedback. This study demonstrated that shoulder external rotation exercise in side-lying with visual EMG biofeedback is a clinically effective training method for reducing posterior deltoid activity and increasing infraspinatus activity.
